Why Tackling Neurodegeneration Requires Global Partnerships
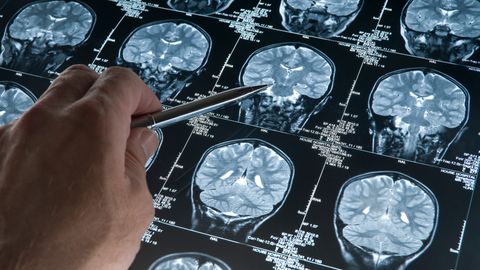
As the global population ages, the prevalence of neurodegenerative diseases is increasing, affecting millions of people around the globe.

Complete the form below to unlock access to ALL audio articles.
Neurodegenerative diseases (NDs) are characterized by synapse dysfunction and the aggregation of proteins in the brain, which ultimately lead to neuronal loss. These brain anomalies affect multiple functions such as speech, movement, balance, memory and cognitive abilities. For most NDs, age is the most common risk factor; however, there are also genetic and environmental factors that contribute to the development of these diseases.1,2 As the global population ages, the prevalence of NDs is increasing, affecting millions of people around the globe, with Alzheimer’s disease (AD) and Parkinson’s disease (PD) standing as the most common conditions.3 Worldwide, it is estimated that approximately 50 million people are living with AD while more than 10 million people suffer from PD.4,5
Unfortunately, a comprehensive understanding of the mechanisms behind these disorders remains elusive. We still lack reliable biomarkers for early diagnosis, let alone effective treatments. However, the advent of high-throughput genomics and proteomics methods promises to change this. These technologies enable researchers to investigate the complex molecular underpinnings of these conditions on an unprecedented scale. By analyzing large datasets of genetic and proteomic information, scientists can identify genetic variants and protein profiles related to these disorders. This, in turn, facilitates the development of diagnostic tools and the discovery of therapeutic targets that could help to stop disease progression.
Exploring protective genes using GWAS
Genome-wide association studies (GWAS) scan the genome of large groups of people for small genetic variants called single nucleotide polymorphisms (SNPs).6 When comparing the genome of patients with controls, these markers can pinpoint genes that are likely involved in disease development.6 Several studies have found that both AD and PD have genome-wide associations in the highly polymorphic human leukocyte antigen (HLA) locus located on human chromosome 6.7,8,9 HLA comprises a large set of genes encoding cell-surface proteins that present antigens to the immune system. “HLA proteins work by recognizing pieces of self and non-self – usually antigens derived from pathogens – and presenting those pieces to the immune system to produce an immune response. Our HLA genes determine which antigens are presented to our immune system,” explains Dr. Emmanuel Mignot, professor in the Department of Psychiatry and Behavioral Sciences at Stanford University. He led a team that analyzed HLA loci in more than 170,000 patients with PD and AD and compared them with healthy controls.9 The team found that people carrying a particular allele of the HLA complex called DR4 are 10% less likely to develop AD and PD. “I was surprised to find a common genetic factor across these non-genetic diseases as it suggests that there is a common mechanism across these conditions,” says Mignot. Further analysis of these patients revealed that people carrying the DR4 allele had a later onset of symptoms and fewer neurofibrillary tangles. These tangles – very common in AD – are long, filamentous aggregates of a protein called tau. “Tau tangles seem to be a response to neuronal injury; they happen after head trauma, for example. Further, tau aggregates are found in several other NDs and even in some autoimmune encephalitis. Tau is also likely to be a catalyst for some other forms of NDs such as PD and amyotrophic lateral sclerosis (ALS). As soon as neuronal injury occurs tau gets involved and may make the situation worse," Mignot explains. “So, tau was a logical candidate, and indeed, we found that DR4 was associated with less tau pathology.”
To better understand the mechanism behind this protective effect, the researchers tested how DR4 binds to different segments of the tau protein. Interestingly, they found that DR4 recognizes a particular tau segment (called PHF6) that is associated with aggregation. “Our current hypothesis is that DR4 recognizes and presents this particular segment of tau, generating an immune response that can reduce aggregation and slow down the progression of these diseases,” explains Mignot. This opens the possibility for the development of a vaccine that could help those people carrying the protective DR4 gene (around 30% of Caucasian populations). “We need to test this first in animals. It’s not perfect, but relatively straightforward. You can inject this particular segment of tau into mice that carry the DR4 gene and see if this triggers an immune response that slows down the aggregation of tau.” If this works, the next step will be to test the vaccine in humans.
This study was the result of a big collaborative effort that involved more than 160 researchers from 25 countries and combined dozens of medical and genetic databases from patients of different ethnic backgrounds in Europe, East Asia, the Middle East and the Americas. “I'm a big collaborator. I like to say that I'm a beggar. I love to collaborate with people, to get people to work together. It took a lot of work to convince all these people to contribute their data. But I think it's really important to study these diseases across different ethnic groups. It's not only the right thing to do because you want your finding to benefit all of mankind, but also because scientifically, it helps you to pinpoint the genes that are involved in a much more certain manner making your results more robust and gaining more insights into the underlying mechanisms,” emphasized Mignot.
Proteomic analysis for early diagnosis
Diagnosis of most neurodegenerative diseases is challenging as it is based on clinical tests that do not always discriminate between similar disorders. Moreover, the development of many neurodegenerative diseases can begin 10–20 years before the clinical symptoms are evident.5,10 This means that by the time a patient is diagnosed with the disease, the level of neurodegeneration is already severe. It is critical to find new diagnostic methods and biomarkers that allow earlier and more accurate disease detection to improve treatment strategies. “Biomarker discovery is a very difficult field because you need to involve different fields of expertise,” explains Dr. W. Andy Tao, professor of analytical chemistry and biochemistry at Purdue University. Tao formed part of a multidisciplinary team that applied a proteomics strategy to uncover diagnostic biomarkers for PD from urinary extracellular vesicles (EVs).11
EVs are membrane-encapsulated vesicles secreted by cells and involved in intercellular communication.12 Several studies showed that potential biomarkers for PD can be identified in cerebrospinal fluid, saliva and plasma-derived EVs.13,14,15 However, the isolation of EVs from these biofluids is often challenging due to the low yield and the presence of impurities. In 2018, Tao developed a method that allows for the highly efficient capture of EVs from urine samples.16 “The method uses magnetic beads and is much more easy, fast and reproducible than the standard ultracentrifugation,” explains Tao. “In that paper, we did a phosphoproteomics analysis on these EVs and found an important PD-linked phosphorylated protein.” Researchers working at the Michael J Fox Foundation-funded LRRK2 Biomarker Project at Columbia University read that paper and proposed a collaboration with Tao. “They gave us 82 urine samples from PD patients, and we carried out the analysis: we isolated the EVs, performed proteomic and phosphoproteomics analysis and then used another 56 samples for machine learning to sort out a panel of proteins that could potentially be used as markers for PD,” recalls Tao.
This study is a truly interdisciplinary cooperation that includes chemists, biochemists, LC-MS proteomics experts, bioinformatic statisticians, clinicians and neuroscientists. As biomarkers previously thought to be undetectable in the urine become uncovered, this type of non-invasive testing opens a new window in early diagnostic development. Tao thinks that the same methodology could be eventually used as an early diagnostic tool for other NDs as well as cancer. “Biomarker discovery always comes with a lot of challenges; but I truly believe that we have made great progress, that will eventually lead to a real clinical application. I’m optimistic,” he concludes.
Collaboration is the key
After several decades of intense research, we still don’t have an effective treatment for AD and PD. “These are not simple diseases; there’s much more heterogeneity than we suspected,” Mignot explains. Thus, the knowledge for potential treatment builds up with every new piece of research. “I think we're going to conquer this one stone at a time,” reflects Mignot. It is only through collaboration that this will effectively happen. Much of the recent progress in ND research heavily relies on collaborative efforts and data-sharing initiatives. Researchers worldwide from different fields – including genetics, proteomics, brain imaging and clinical medicine – pool their data and findings through consortiums, such as the Alzheimer's Disease Neuroimaging Initiative (ADNI) and the Parkinson's Progression Markers Initiative (PPMI). The complexity of the data generated by these large-scale studies also drives the development of new bioinformatic and artificial intelligence tools. This holistic approach enhances our understanding of the mechanisms underlying these devastating conditions, offering hope for more effective treatments and diagnostic methods.
About the interviewees:
Emmanuel Mignot is the Craig Reynolds Professor of Sleep Medicine in the Department of Psychiatry and Behavioral Sciences at Stanford University and the Director of the Stanford Center for Narcolepsy. His research focuses on the neurobiology, genetics and immunology of narcolepsy, with indirect interest in the neuroimmunology of other brain disorders such as autoimmune encephalitis or neurological paraneoplastic syndromes.
W. Andy Tao is a professor of analytical chemistry and biochemistry at Purdue University. His research focuses on the development of technologies to target and discover novel biomarkers for different diseases.